User:Mr. Ibrahem/Oxcarbazepine
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ɒks.kɑːrˈbæz.ɪˌpiːn/ |
| Trade names | Trileptal, Oxtellar XR, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601245 |
| License data | |
| Routes of administration | By mouth (tablets or liquid)[1] |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | >95% |
| Metabolism | Liver (cytosolic enzymes and glucuronic acid) |
| Elimination half-life | 1–5 hours (healthy adults) |
| Excretion | Kidney (<1%)[1] |
| Identifiers | |
| |
| Chemical and physical data | |
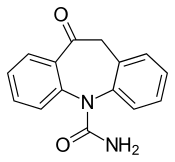
| Formula | C15H12N2O2 |
| Molar mass | 252.273 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Oxcarbazepine, sold under the brand name Trileptal among others, is a medication used to treat epilepsy and bipolar disorder.[2] For epilepsy it is used for both focal seizures and generalized seizures.[5] It has been used both alone and as add-on therapy in people with bipolar who have had no success with other treatments.[6][2] It is taken by mouth.[2]
Common side effects include nausea, vomiting, dizziness, drowsiness, double vision and trouble with walking.[3] Serious side effects may include anaphylaxis, liver problems, pancreatitis, suicide, and an abnormal heart beat.[3][5] While use during pregnancy may harm the baby, use may be less risky than having a seizure.[7] Use is not recommended during breastfeeding.[7] In those with an allergy to carbamazepine there is a 25% risk of problems with oxcarbazepine.[3] How it works is not entirely clear.[2]
Oxcarbazepine was patented in 1969 and came into medical use in 1990.[8] It is available as a generic medication.[5] A month supply in the United Kingdom costs the NHS about £6.50 as of 2019.[5] In the United States the wholesale cost of this amount is about $7.15.[9] In 2017, it was the 207th most commonly prescribed medication in the United States, with more than two million prescriptions.[10][11]
References[edit]
- ^ a b "Oxcarbazepine Drug Label". Archived from the original on 2015-12-08. Retrieved 2015-11-04.
- ^ a b c d e "Oxcarbazepine Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 30 March 2019. Retrieved 13 April 2019.
- ^ a b c d "DailyMed - OXCARBAZEPINE- oxcarbazepine tablet". dailymed.nlm.nih.gov. Archived from the original on 2015-12-08. Retrieved 2015-11-11.
- ^ "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 29 September 2020. Retrieved 9 September 2020.
- ^ a b c d British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. pp. 319–320. ISBN 9780857113382.
- ^ Mazza M, Di Nicola M, Martinotti G, Taranto C, Pozzi G, Conte G, Janiri L, Bria P, Mazza S (April 2007). "Oxcarbazepine in bipolar disorder: a critical review of the literature". Expert Opinion on Pharmacotherapy. 8 (5): 649–56. doi:10.1517/14656566.8.5.649. PMID 17376019.
- ^ a b "Oxcarbazepine Use During Pregnancy". Drugs.com. Archived from the original on 13 April 2019. Retrieved 13 April 2019.
- ^ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 532. ISBN 9783527607495. Archived from the original on 2019-03-06. Retrieved 2019-03-02.
- ^ "NADAC as of 2019-02-27". Centers for Medicare and Medicaid Services. Archived from the original on 2019-03-06. Retrieved 3 March 2019.
- ^ "The Top 300 of 2020". ClinCalc. Archived from the original on 12 February 2021. Retrieved 11 April 2020.
- ^ "Oxcarbazepine - Drug Usage Statistics". ClinCalc. Archived from the original on 28 February 2020. Retrieved 11 April 2020.
