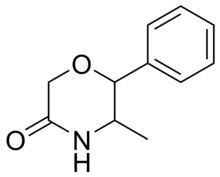
Fenmetramide
From Wikipedia, the free encyclopedia
Chemical compound
Not to be confused with Fenmetozole.
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C11H13NO2 |
| Molar mass | 191.230 g·mol−1 |
| 3D model (JSmol) | |
| |
Fenmetramide is a drug which was patented as an antidepressant by McNeil Laboratories in the 1960s.[1] The drug was never marketed.[2] It is the 5-ketone derivative of phenmetrazine and would similarly be expected to produce psychostimulant effects, though pharmacological data is lacking.
Synthesis[edit]

Ex 1: dl-Norephedrine Fb: [14838-15-4] (HCl): [154-41-6] was treated with NaH in mineral oil and later reacted with the ethyl chloroacetate [105-39-5]. Cis-product
Ex 2: dl-norpseudoephedrine was used => trans-product.
See also[edit]
- Fenbutrazate
- Phendimetrazine
- Phenmetrazine
- Flumetramide (Skeletal muscle relaxant)
References[edit]
- ^ a b US 3308121, Gannon WF, Poos GI, "3-Morpholinones, thiones and 5,6-oxazines", issued 7 March 1967, assigned to McNeil Laboratories
- ^ Triggle DJ (1997). Dictionary of pharmacological agents. London: Chapman & Hall. ISBN 978-0-412-46630-4.
- ^ Clarke, Frank H. (1962). "cis- and trans-3-Methyl-2-phenylmorpholine". The Journal of Organic Chemistry 27 (9): 3251–3253. doi:10.1021/jo01056a063.
- ^ Spassov, S. L., Stefanovsky, J. N., Kurtev, B. J., Fodor, G. (August 1972). "Optisches Drehungsvermögen und Konformation, VI. Protonenresonanzspektren und Konformation einiger stereoisomerer 2.3‐disubstituierter Morpholine und 5‐Oxo‐morpholine". Chemische Berichte. 105 (8): 2467–2475. doi:10.1002/cber.19721050807.
- ^ W. Klyne, (the late), Scopes, P. M., Berova, N., Stefanovsky, J., Kurtev, B. J. (January 1979). "The circular dichroism of some derivatives of tetrahydro-1,3-oxazine, tetrahydro-1,4-oxazine(morpholine)and 1,3-oxazolidine". Tetrahedron. 35 (17): 2009–2012. doi:10.1016/S0040-4020(01)88970-5.
| |||||||||||||||||||||
| |||||||||||||||||||||
| |||||||||||||||||||||
| |||||||||||||||||||||
| |||||||||||||||||||||
